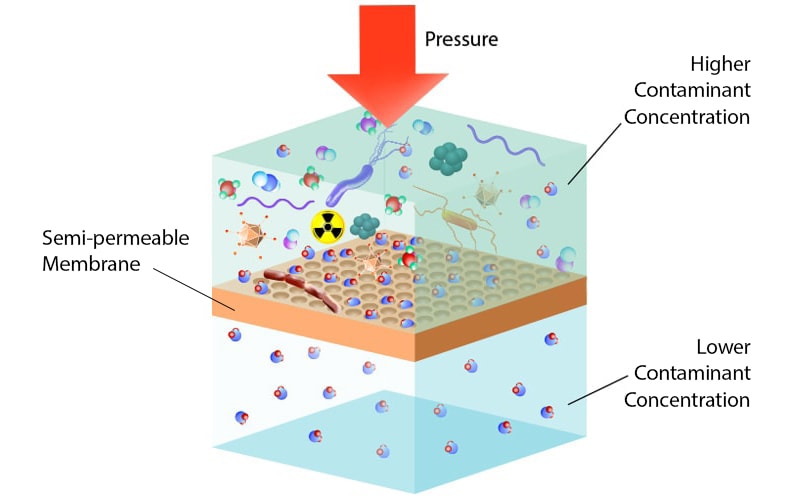

Osmosis shoots up in a condition where the difference in concentration of solute across the membrane is higher.As the concentration of solute molecules is essential in the driving force of osmosis, any changes in the concentration directly affect the rate of osmosis.As the molecules become more energetic, their movement also increases, and thus the process of osmosis is escalated.This occurs because, with the increase in temperature, the energy of the molecules also increases.The rate of osmosis increase with the increase in temperature of the system.Osmosis is a result of various factors, and thus the rate of osmosis is influenced by a number of such factors: Temperature This process continues until the pressure on both sides becomes equal leading to equilibrium. This pressure results in the forcing of water across the membrane.As a result, the water molecules in the pure water exert more pressure towards the solution with lower solvent concentration. The interaction between the solute and water molecules reduces the pressure exerted by the water molecules in the solute solution.The chemical potential of pure water in one solution differs from the chemical potential of water in the solution with solute molecules.Therefore, the process of osmosis has been explained with the concept of chemical potential.This theorem too doesn’t hold true as the size of the solute molecules does not influence the movement of a solvent across the membrane. Another theorem put forth by several other scientists is the fact that the solute molecules in the solution attract the solvent molecules across the membrane.This theorem, however, doesn’t explain the process of reverse osmosis, which occurs against the concentration gradient with the movement of solvent towards the solution of lower concentration.One such theorem explains that the movement of water across the membrane results due to the concentration gradient of water in the two solutions. Several descriptions have been made to understand the driving force of osmosis.According to the definition of osmosis, in such a case, pure water moves across the membrane towards the solute solution.One of the solutions is pure water while the other one is a solution of some solute and water.In order to describe the process and mechanism of osmosis, we take two solutions separated by a semi-permeable membrane.Mechanisms of osmosis (How does osmosis work?)įigure: A schematic diagram showing how osmosis works.
